The Discovery
The Alkaline earth metals were discovered by Sir Humphry Barthlolomew Davy, besides radium (discovered by Marie Curie) and Baryllium (discovered by Nicholas Louis Vauguelin). Davy's path to discovering the Alkaline earth metals was paved by his background in working in an apothocary, although he had apprenticed as a surgeon, and discoveries made before his time.He was the first to isolate several of the alkaline earth metals from the alkali metals. Davy was able to isolate calcium by using a mixture of lime (CaO) and mercuric ocide (HgO). Davy did this with each of the alkaline earth metals he studied. Marie Curie was born into learning about science, her father was a phisycs teacher. Curie also had the aid of her husband in the discovery of her elements.
Unique Properties
Alkaline earth metals share many traits with metals. Uniquely, they have two electrons on the outer shell. Although alkaline earths share many traits with the alkali metals, alkaline earths have a smaller atomic radii because the two valeance electons are not as tightly bonded with the nucleus as they are in alkali metals. Alkaline earth metals are also very reactant (mostly with nonmetals), so they are difficult to get a hold of even though they make up such a large part of the earth's crust.Alkaline earth metals also react with water to produce a hydrogen gas. Magnesium in its purest form is combustable and will react with oxygen to produce a white light. Most alkaline earths are shiny, and most are silver or white. When heated, they glow different colors. They are soft, but unlike alkali metals they are not soft enough to be cut with a knife. Alkaline earth metals act as bases (like most metals).
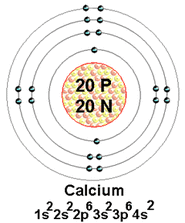
Calcium is an alkaline earth metal. It's melting point is 1542.2 °F and boiling point is 2703.2 °F. It's a silver color. The density of calcium is 1.55 g/cm3. Calcium is used in bone structures and the sturcture of other types of shells. It can be obtained from chalk, limestone, marble, and is found in 3.5% of the crust.
Uses
In WW2 magnesium was heavily used in air craft materials. Magnesium is also ranked number in seven in the elements found in the human body. Calcium is used in the structure of lifeforms and other shells.